Test Complete
- Questions
- Score
- Minutes
| Overall Results | |
|---|---|
| Total Questions |
| Category Results | |
|---|---|
Blind Insertion Airway Devices (BIAD)
Category: Airway
Topic: Advanced Airway Management
Level: AEMT
Next Unit: Assessing Difficult Airways
16 minute read
Blind Insertion Airway Devices (BIADs)
With the importance of maintaining the airway and the sheer number of patients who experience airway compromise, several companies have created devices to ease the process of obtaining a "definitive airway." A definitive airway is one that cannot be easily compromised by vomit, secretions, or swelling. (e.g., a tube of plastic that maintains a path for air at all costs).
In the past, this role was traditionally played by intubation and placement of an endotracheal tube. However, the complexity of this procedure leads to frequent failures in the uncontrolled EMS environment. BIADs are tools that simplify this process. Per the name, they can be inserted blindly without visualization of the vocal cords to confirm placement.
This section will review the indications for BIAD use, the two most common types, and the usage of BIADS in general.
Indications, Contraindications, and Complications
INDICATIONS: BIADs are used when a definitive/advanced airway is required, and intubation is either unsuccessful or unrealistic. The indications for an advanced airway are as follows:
- An unresponsive patient who is hypoxic despite adequate BVM ventilation
- Patient with trauma to the face/neck who is experiencing confirmed or suspected swelling.
- Penetrating injury to the head or neck
- More routine injuries with suspected long transport times
CONTRAINDICATIONS: The contraindications to BIAD are focused on causing vomiting, bleeding, or potentially perforating the larynx/esophagus. They are as follows:
- An intact gag reflex
- Known esophageal disease (cancer, varices, stricture)
Cancer tissue is very fragile--you can easily perforate the esophagus (or trachea in laryngeal or tracheal cancer) without feeling the breach of resistance!
- Laryngectomy with a stoma
- Caustic ingestion or airway burns
- Stature < 5 feet
COMPLICATIONS: The complications of BIAD use surround the nature of pushing a rigid tube into relatively soft tissue and inflating a balloon within it (trauma/compression), along with the risk of inflating the stomach.
- Inflation of the stomach leads to regurgitation and aspiration of gastric contents
- compression of vascular structures in the neck
- Trauma to the airway/esophagus
- nerve injury to the "laryngeal nerves" that control the vocal cords.
- Hypotension due to positive pressure ventilation and/or vagus nerve stimulation.
Supraglottic Airways
The Combitube used to be the most common BIAD in the prehospital setting. However, thanks to advances in technology and better data, that device has been replaced by other supraglottic airways (SGAs) like the King LT airway and the i-Gel. Both of these airways are dual-lumen devices. One of the key differences between the Combitube versus the King LT is that the King LT only has one inflation cuff, whereas the Combitube has two. The i-Gel has neither and acts similarly to that of a laryngeal mask airway (LMA), which we will discuss in the next section of this unit.
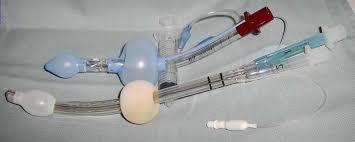
The King LT supraglottic airway is designed for blind insertion into the esophagus. It has two inflatable cuffs: a distal cuff that seals off the esophagus and a proximal cuff that seals the oropharynx just above the laryngeal inlet. Ventilation is delivered through a single lumen with ports located between the cuffs, directing air into the trachea. Proper placement is confirmed by observing chest rise and listening for bilateral breath sounds.
Unlike the Combitube, the King LT's design precludes tracheal insertion. If it were inserted into the trachea, ventilation would be blocked due to the closed distal end of the tube. The balloons are inflated simultaneously to create effective seals, allowing positive pressure ventilation and providing a port for gastric tube insertion to help decompress the stomach.
In cases where ventilation is difficult or breath sounds are absent, the device may need slight repositioning. As with any supraglottic airway device, factors such as bilateral pneumothorax can complicate placement confirmation by auscultation, making capnography an essential adjunct for verifying proper ventilation.
Laryngeal Masks
The laryngeal mask airway (LMA) is an ET tube that goes over the larynx instead of into it and primarily used in the hospital environment. The King LT and the i-gel are the go-to in the prehospital realm.
 The i-gel has replaced the LMA in the prehospital setting and is also placed blindly. After figuring the patient's weight, it is advanced into the oropharynx until resistance is met. The i-gel seals as the patient's body temperature warms the gel-type distal end of the device and provides a custom fit to protect the airway during ventilation. Correct placement will position it to sit atop the glottis and protect the airway from vomit/blood/secretions. Auscultation for breath sounds and end-tidal capnography is the preferred method of confirming placement.
The i-gel has replaced the LMA in the prehospital setting and is also placed blindly. After figuring the patient's weight, it is advanced into the oropharynx until resistance is met. The i-gel seals as the patient's body temperature warms the gel-type distal end of the device and provides a custom fit to protect the airway during ventilation. Correct placement will position it to sit atop the glottis and protect the airway from vomit/blood/secretions. Auscultation for breath sounds and end-tidal capnography is the preferred method of confirming placement.
Usage of BIADS
To place any BIAD:
- Ensure the patient is pre-oxygenated with 100% O2 for at least 30 seconds.
- Assemble your equipment: Suctioning, lubricant, oxygen, BVM, and at least two full correctly-sized BIAD devices.
- Check the equipment to ensure it is in working order.
(If the BIAD of choice is the King LTD airway, ensure the cuffs will inflate to the proper pressure.) - Lubricate the distal tip of the BIAD and open the patient's airway.
- Remove any adjunct (OPA, NPA) present and suction the airway of any secretions.
- Insert the BIAD at a 90º angle (pointing to the right or left corner of the mouth) with the distal tip sliding back over the patient's tongue.
- Rotate the BIAD to be in line with the patient's airway.
- The device is advanced until the phalange rests against the patient's teeth.
- Inflate the pilot balloon with air from the included syringe to the manufacturer-indicated pressure.
-
Confirm placement:
-
Attach to capnography or capnometry and connect to the BVM.
-
Auscultate over the epigastrium during initial ventilation. If breath sounds are heard in the abdomen, adjust and reassess.
-
Move up to auscultate the left and right lung fields, ensuring equal breath sounds.
-
-
If you hear breath sounds in only one lung (unilateral ventilation), slightly pull back the device while keeping the cuffs inflated. Then, reassess breath sounds.
-
Secure the airway per local protocol and monitor SpO₂ and ETCO₂ continuously.
-
Pneumothorax can cause absent or unequal breath sounds even with proper placement.
-
Always use water-based lubricants such as KY Jelly; never petroleum-based products.
-
Follow manufacturer height and size guidelines (e.g., the King LT is typically for patients over 4 ft tall).