Test Complete
- Questions
- Score
- Minutes
| Overall Results | |
|---|---|
| Total Questions |
| Category Results | |
|---|---|
Acid-Base Balance
Category: Medical
Topic: Acid-Base Balance
Level: AEMT
Next Unit: Carbon Dioxide and pH
15 minute read
Acid-Base Balance
pH
pH is a measurement of acidity. It just measures how acidic something is. When referencing acidity, we're talking about excess hydrogen ions (H+), but in the real world, we're just going to use it as if it were like a "percentage of acids" or, better, the "amount of acidity."
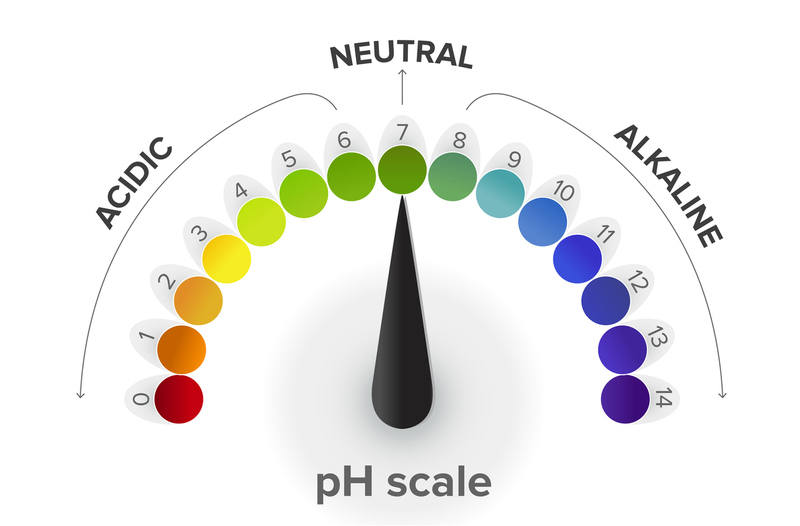
The pH scale goes from 0-14, so neutral is 7 (water is neutral). In the human body, abnormal pH lies on either side of the 7.35-7.45 range:
ACIDOTIC ← 7.35 - 7.45 → ALKALOTIC
The metabolism of homeostasis requires that the pH remains in a narrow "Goldilocks" range of 7.35-7.45. Since physiology is biochemistry, all of the chemical reactions in the human body are affected by pH. Alterations in the pH, resulting in either acidosis (<7.35) or alkalosis (>7.45), can become problematic according to the spectra of illnesses and their severity.
The body needs a pH of 7.35-7.45 to maintain homeostasis. > 0.5 away from neutral in either direction is not conducive to life. So, when we find someone whose acid levels are not conducive to life, we need to counteract the problem to restore homeostasis. The human being requires it to be neither too acidic nor too alkaline but balanced, which is all Acid-Base balance means.
ACIDIC - TOO MUCH ACID - Lower than 7.35 means that there is too much acid in your system.
NORMAL - The "Goldilocks" zone: just right! pH between 7.35 and 7.45 is perfect for homeostasis.
ALKALINE - NOT ENOUGH ACID - or alternately, too much alkali. pH higher than 7.45 is considered alkaline. During homeostasis, the body corrects any imbalances in pH levels through the correction of acidosis and alkalosis.
The normal body pH values between 7.35 and 7.45 are never constant but vary subtly as homeostasis continues. In the pathological range, there exist.
- Respiratory acidosis and alkalosis, and
- Metabolic acidosis and alkalosis.
Homeostasis demands a rapid resolution of all types of acidity and alkalinity, so both have life-threatening consequences if not corrected adequately and correctly.
Daily Acidosis and Homeostasis
Daily acidosis is business as usual! Human beings run on fuel, and human metabolism creates wastes, which are usually acidic at the cellular level. We, therefore, have a tendency toward acidosis at all times. Every day large amounts of acids are generated from metabolism. These acids must be
- excreted,
- metabolized/neutralized to non-charged neutral molecules and/or
- buffered to avoid fatal acidemia.
Homeostasis--with several mechanisms to reduce acids--is responsible.
The acids of metabolism comprise 3 classes:
- Carbonic acid (H2CO3), from the CO2 produced with each breath combined with water. CO2 is removed by pulmonary excretion.
- Organic acids (lactic acid, citric acid) from reactions in day-to-day metabolism. They are eliminated by metabolism. That is, these acids are metabolized to neutral products such as glucose or to CO2 and water.
- Nonvolatile acids, such as sulfuric acids, form metabolism of sulfur-containing amino acids in the diet. They are excreted by the kidneys.
Therefore, acid-base balance is maintained (mainly, but not exclusively) by
- the lungs (removing CO2),
- the liver (metabolism of organic acids), and
- the kidneys (excretion of nonvolatile acids).
Metabolic Acidosis and Alkalosis
►METABOLIC ACIDOSIS: Lactic acidosis occurs when lactic acid production exceeds lactic acid clearance or if metabolism fails to neutralize the amounts normally produced daily (increased production vs. decreased metabolism). (pH < 7.35)
Causes of Lactic Acidosis:
Most cases of lactic acidosis are due to
- marked tissue hypoperfusion from hypovolemia,
- cardiac failure,
- sepsis, or
- cardiopulmonary arrest.
Alternately,
- toxic impairment of cellular metabolism and ischemia can cause it, too. (Diabetes.)
- Alcoholism (hepatic dysfunction).
Response to acidosis: In the event of metabolic acidosis, renal and respiratory responses are mediated by pH changes in the sensory and regulatory cells or the renal tubules and the respiratory center (kidney and lung response).
The inability of respiratory response from respiratory or neurologic disease or before there has been adequate time for respiratory compensation to fully act can fail to remedy the acidosis.
►METABOLIC ALKALOSIS: a condition in which the body produces too much bicarbonate, raising the pH to a level above normal. (>7.45)
Causes of Metabolic Alkalosis:
- Loss of hydrogen ions from the gastrointestinal tract (gastric or diarrhea) or in the urine.
- Hydrogen ion movement into cells.
- Increased serum bicarbonate concentration from alkali administration (antacids)
- Extracellular fluid volume contraction around a relatively constant amount of extracellular bicarbonate, as with dehydration.
Most patients with normal volume and renal function can excrete excess bicarbonate in the urine. For metabolic alkalosis to persist, there must be enough kidney dysfunction to reduce the excretion of excess bicarbonate into the urine.
Respiratory Acidosis and Alkalosis
►RESPIRATORY ACIDOSIS: a condition in which the lungs cannot remove enough of the carbon dioxide produced by the body and is often caused by hypoventilation. Respiratory acidosis is an increase in carbon dioxide levels with a respiratory etiology.
Renal compensation to respiratory disorders takes 3-5 days to complete, so the primary respiratory disorders can be further divided into
- acute respiratory acidosis, and
- chronic respiratory acidosis.
Causes of Respiratory Acidosis:
- Lung disease (COPD, emphysema, pneumonia, etc.)
- Drowning or asphyxiation.
- Sleep apnea.
- Opioid overdose.
►RESPIRATORY ALKALOSIS is the reverse condition, in which the lungs are removing too much carbon dioxide from the body, increasing the pH level to a more alkalotic level than normal and is often caused by hyperventilation.
Causes of Respiratory Alkalosis:
- Hyperventilation.
Renal compensation to respiratory disorders takes 3-5 days to complete, so the primary respiratory disorders can be further divided into
- acute respiratory alkalosis, and
- chronic respiratory alkalosis.